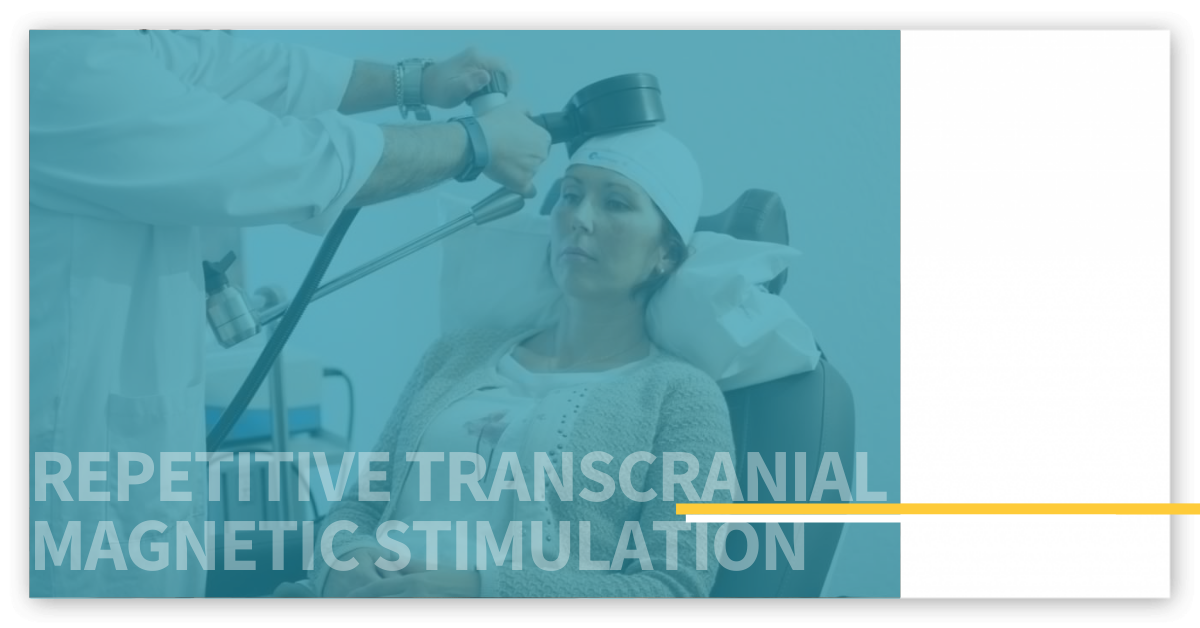
What is Repetitive Transcranial Magnetic Stimulation (rTMS)?
LISTEN TO THIS ARTICLE:
What it is
Transcranial magnetic stimulation (TMS) began as a form of brain mapping. Researchers create maps of brain surface activity using single-pulse and paired-pulse TMS.
When the stimulation happens sequentially, that’s called repetitive TMS (rTMS). Over the long term, rTMS can alter brain chemistry and lead to therapeutic benefits.
Who needs it
In 2008, the U.S. Food and Drug Administration cleared repetitive transcranial magnetic stimulation for treating major depressive episodes that have failed to respond to at least one antidepressant. Studies dating back to 1985 suggest that people with psychosis and anxiety may also benefit from rTMS.
Patients with magnet sensitivities due to implanted devices should consider rTMS only if there’s a compelling medical reason to do so.
How it works
Repetitive transcranial magnetic stimulation employs a wand outfitted with small coils that deliver multiple magnetic pulses in quick succession. As the pulses pass through the skull into the targeted region, they create magnetic field (comparable to an MRI scan). In turn, this makes small electrical currents that stimulate nerve cells.
When placed over the patient’s forehead, the coils focus the magnetic field on the dorsolateral prefrontal cortex (DLPFC), an area of the brain that functions abnormally in major depressive disorder.
The beauty of rTMS lies in its ability to pinpoint a specific area of the brain, which helps reduce the risk of side effects. Scientists continue to debate which area is best but for now the DLPFC has been established as the locus of treatment for depression.
Where to go
Since rTMS requires no anesthesia, a specially trained nurse can perform the procedure in an office setting under a psychiatrist’s supervision.
What to expect
The typical rTMS session takes 30-40 minutes. Treatments are scheduled five days a week for at least three weeks, but can last longer as needed.
Patients report mild sensations of knocking or tapping during rTMS therapy.
Although rTMS is less effective than ECT, large studies have shown that 20-30 percent of patients experience remission by the end of treatment.
What could happen
Seizure is a very rare, but possible, side effect of rTMS. Therefore, the treatment team must be ready to administer anti-seizure medication.
Patients can discuss with their doctor the option of taking a long-acting benzodiazepine before the procedure to reduce anxiety and prevent seizures.
Mild headaches or lightheadedness, muscle twitching, and head pain near the coil location are the most common side effects.
Long-term side effects are unknown because rTMS is a relatively young technology. And even though it’s FDA-cleared, insurance may not cover rTMS therapy. Some plans require that physicians seek pre-approval before they will pay for the procedure.


 Learn
Learn Find Help
Find Help